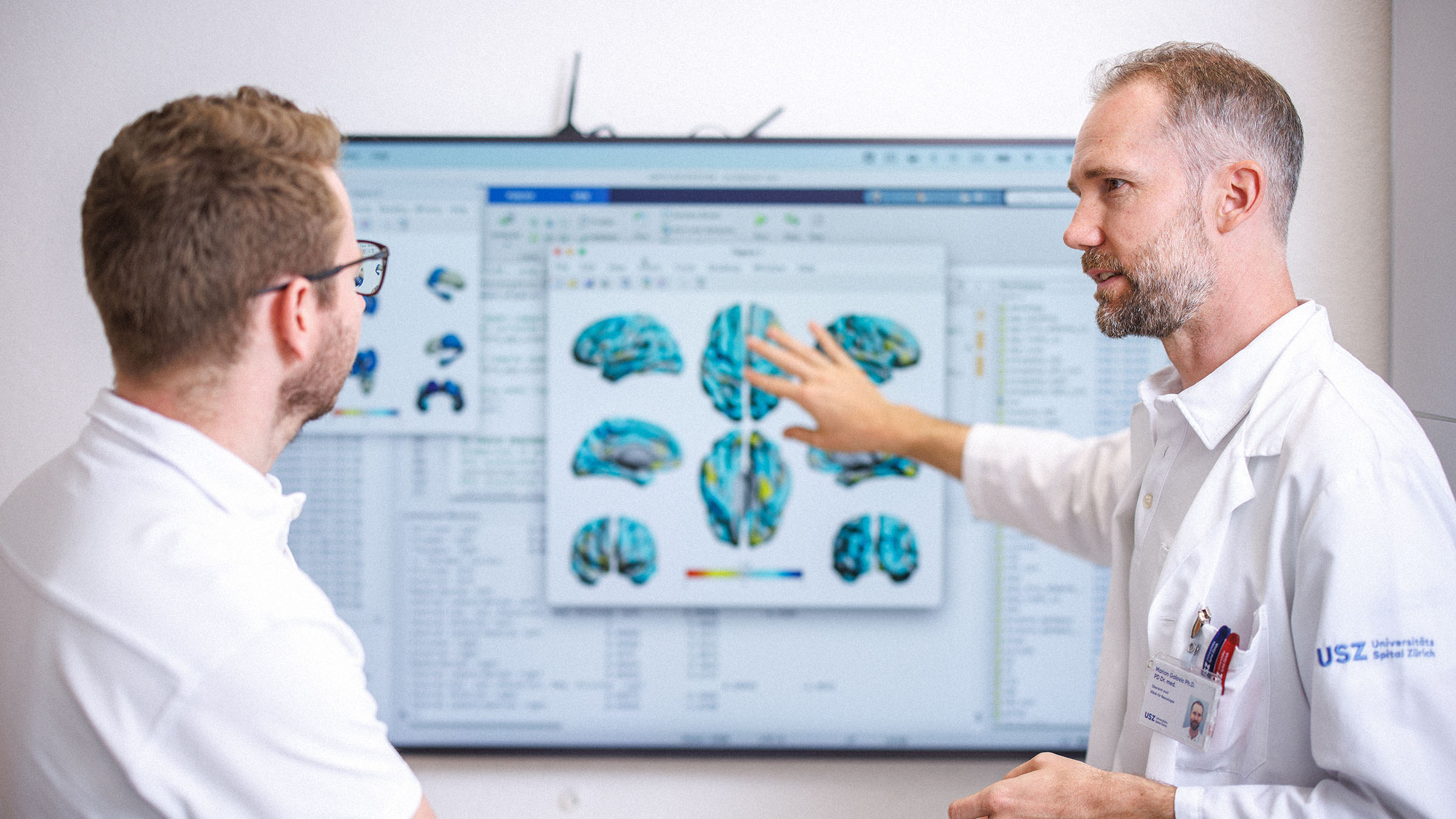
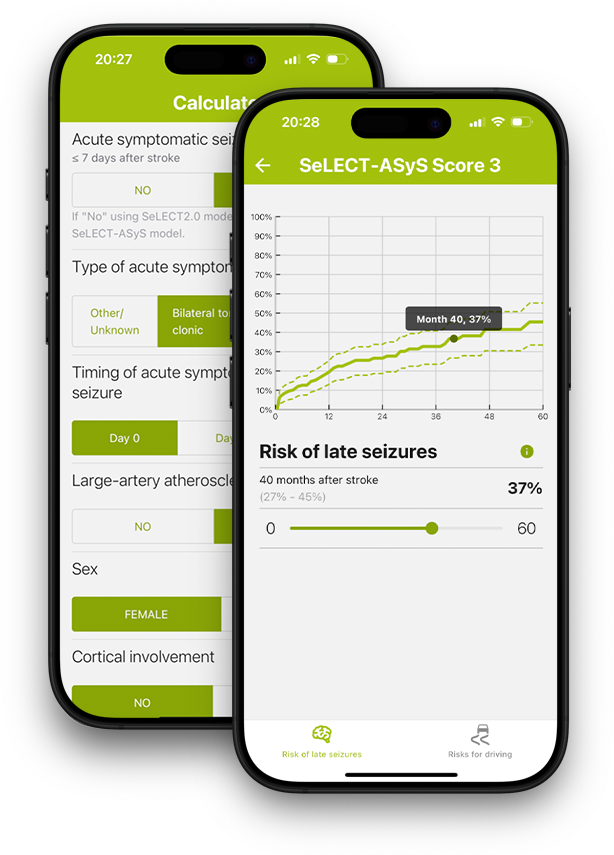
Predicting Epilepsy
After Stroke.
International multicentre prospective cohort building a harmonised, biomarker-informed, trial-ready platform for post-stroke epilepsy prediction and prevention research — coordinated by University Hospital Zurich.